Herd Immunity and SARS-CoV-2
A Political Play in 7 Parts (if Parts=Lies): Parts 6 (cont'd), 7, & Conclusion
Preface to these parts:
Keep in mind that the following continues the distillation of my thought process and discovery, detailing what I was uncovering back in August and September (then written in October).
Some of these things have come out. Some of these things are known already to whoever is reading this. At the time though, they were not widely known or recognized. Further, they were actively ignored and denied.
Part 6 cont’d:
What they knew beforehand is only further supported by what we know now
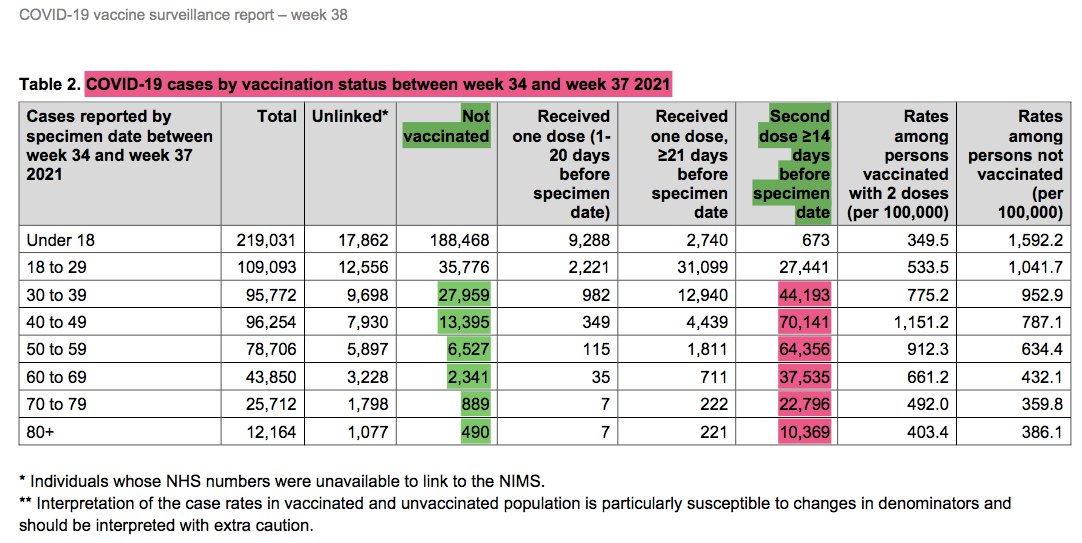
This is not a pandemic of the unvaccinated. Yet the message still clings to the falsehood that somehow getting these vaccines can protect anyone other than the individual. One only had to look outside of the U.S. to see the contradiction. Take data out of Israel, Singapore, Iceland (from Johns Hopkins University Center for Systems Science and Engineering (JHU CSSE)1), and the UK2 for example. Some of the most highly vaccinated nations (>70%) recently have recorded their highest case rates since the pandemic began. The following data are a clear indication that infections, hospitalizations, and deaths still occur and at high levels in the vaccinated populations, not just the rarity they wish it was.
Viewed on Oct. 20, 2021 for comparison to Oct. 20, 2020:
Iceland:
Here’s most current:
Singapore:
Israel:
Here’s most current:
UK data:
UK data put another way:
This highlights that the vaccinated account for a significant portion of those affected regardless of age group. In none of the above cells do the unvaccinated cases represent 99% of any criterion. In fact, for older age groups a greater proportion of cases, hospital encounters, and deaths occurred in the vaccinated over this timeframe (highlighted in red). The claim that the unvaccinated are straining the healthcare system or whatever other rationalization they want to spout are just factually untrue.
Since younger ages are at lowest risk of severe outcomes, let’s crunch the numbers yet another way:
Age 30-80+: 352,458 total cases; 51,601 unvax (14.6%) vs 249,390 (70.8%) vaxxed
Age 50-80+: 5,589 hospitalized; 1,339 (24.0%) unvax vs 4,063 (72.7%) vaxxed
Age 50-80+: 2,983 deaths w/in 28 days + test; 615 (20.6%) unvax vs 2,239 (75.1%) vaxxed
Age 50-80+: 3,499 deaths w/in 60 days +test; 722 (20.6%) unvax vs 2,619 (74.8%) vaxxed
The percentage of total cases, hosp. or deaths for the vaxxed is nearly the same to the percentage of vaccinated in the respective age groups. This means the vaccine is having nearly zero effect on prevention of infection, hospitalization, or deaths. This is not my assessment. This is the assessment using the CDC’s Vaccine Effectiveness Screening Method. (**See their powerpoint slide & chart below for depiction from U.S. data; they assume a linear relationship between percent vaccinated cases vs. percent population vaccinated for the zero vaccine effectiveness line, for example if 100% of cases are in vaccinated and 100% of population is vaccinated then VE=0. And they apply this for cases, hospitalizations, and deaths.) So if ~80% +/- (depending on age group) of the UK population was vaccinated at this time, then the percent of cases, hospitalizations, and deaths being ~75% means a VERY near zero VE.
NOTE: Continued reports since then have shown similar percentages and totals, with higher numbers crossing into the 40-49 vaxxed group and nearing the 30-39 group.3 This is even with booster percent at ~76%.
Again, this data was from September and accessible publicly. The failures were known or at least should have been. The question again is “Extreme incompetence or lies?” We’ll see in a moment the answer.
A final note regarding the UK data. Interestingly, PHE declared at the end of this report that they will “stop producing these comparisons” because “the state of the actual pandemic and the no-vaccination pandemic scenario have become increasingly less comparable.” Hmm, I wonder why? They say:
“…recent results from the no-vaccination scenario show that the pandemic, in the absence of vaccination and additional interventions, would have peaked due to natural immunity…The largely unmitigated epidemic in the no-vaccination scenario has now reached the end of its epidemic curve, while the real-world pandemic continues and estimates of infections averted will have reached their peak.”
Sounds like a fancy, convoluted way of saying that oops, the real world vaccination program is allowing the pandemic to continue rather than fizzle out. Also a crafty way of admitting that they realize the likelihood that “infections averted” will quickly approach zero, aka everyone will be infected in short order.
Another way to look at the data. The only way for these cases to be as widespread, and in areas where vaccine passports had been in play, is if the vaccines don’t prevent transmission. And obviously don’t prevent infection. Given the increased viral loads with delta variant even in the vaccinated, and the fact that they are more likely to be spreading a variant to which other vaccinated individuals are susceptible (see part 5), make it pretty conclusive that the pandemic is not being curbed by the vaccine. It isn’t a stretch to conclude that the propagation is at least in part due to the vaccine. The Public Health Scotland data4 5from September 11 - October 8, 2021 showed that:
• 54% of COVID-19 cases were among the vaccinated vs. 46% unvaccinated
• 69% of COVID-19 hospitalizations were among the vaccinated vs. 31% unvaccinated
• 82.2% of COVID-19 deaths were among the vaccinated vs. 17.8% unvaccinated
A recent study6 from Israel details a nosocomial outbreak in July among a 96.2% vaccinated population that began in a fully vaccinated patient with spread predominantly among fully vaccinated patients and hospital staff, despite PPE and other mitigating measures. “It challenges the assumption that high universal vaccination rates will lead to herd immunity and prevent COVID-19 outbreaks.” This study also highlights that: 1) increased age and 2) co-morbidities are far more significant when it comes to COVID disease severity, regardless of vaccination status.
Yet these two important facts have been routinely omitted in main stream media and other press briefings regarding COVID-19. The vast majority of deaths from COVID, approximately 95%, were in those with co-morbidities, and on average with at least 4, YES 4, co-morbidities. I quote the CDC:
“For over 5% of these deaths, COVID-19 was the only cause mentioned on the death certificate. For deaths with conditions or causes in addition to COVID-19, on average, there were 4.0 additional conditions or causes per death.” (emphasis added)7
This is nothing new; it’s been known since at least Feb. 2021.8 In tiny print they also clarify:
that the 5% “was likely related to lack of detail listed about other conditions present at the time of death.”9 In essence admitting that the other 5% likely had co-existing conditions as well! Since March 2020 the CDC had a list of such conditions that increase the risk of severe disease10 (similar to what was known about severe SARS and MERS as well). Side note: Influenza is in the most frequently listed…I thought the flu was non-existent this past year??
Concerning older age, 74.5% (current total percentage) to 80.8% (2020 percentage) of deaths from COVID in the U.S. occurred in those 65 years of age and older (9). The starkly skewed preponderance for severe disease and death has been recognized nearly from the start of the pandemic in the U.S. and continues to hold true.
Yet never for nearly 2 years has it been stressed that getting healthier could be an effective way to lower the risk of severe disease. Modifiable risk factors are a common and important concept in medicine. It’s inexcusable, anathematic, and puzzling that they have been ignored for this one disease, yet applied to every other facet of medical care for decades.
Instead for example, a recent study11 showed: for 5-11-year-olds the portion who are overweight or obese “is now 45.7 percent, up from 36.2 percent before the pandemic. The same figures rose by 5.2 percent among 12-15-year olds and 3.1 percent among 16- and 17-year-olds.”12 But I digress.
Even data from the U.S. has become available which shows the real life meaning of waning vaccine effectiveness. It comes from the Joint Artificial Intelligence Center of Department of Defense Humetrix Project Salus.13 14 In FDA VRBPAC meeting documents from Dec. 2020 they highlight that the DOD, CMS, & CDC among others will work with the FDA to analyze COVID data related to the vaccines.15
The following data analysis correlates well with the data coming out of the UK.
MEDICARE DATA:
Effectiveness of mRNA COVID-19 Vaccines Against the Delta Variant Among 5.6M Medicare Beneficiaries 65 Years and Older- Weekly update of September 28, 2021.16:
Feb. 2021 to Aug. 2021, light blue bar represents proportion of hospitalizations for COVID-19 in fully vaccinated individuals:
NOTE ABOVE: clearly the unvaxxed do not make up anything close to 99% (except at the beginning of February when the unvax % (Red Line) was nearly 100%). As early as May 1, 2021 vaccinated individuals make up ~50% of those 65+ hospitalized (and this was when delta only accounted for 4% of sequenced cases, so NOT dominant yet in the slightest and therefore not able to be blamed for the vaccine failure).
Breakthrough cases at least twice as high 5-6 months post-vax vs. 3-4 months post-vax
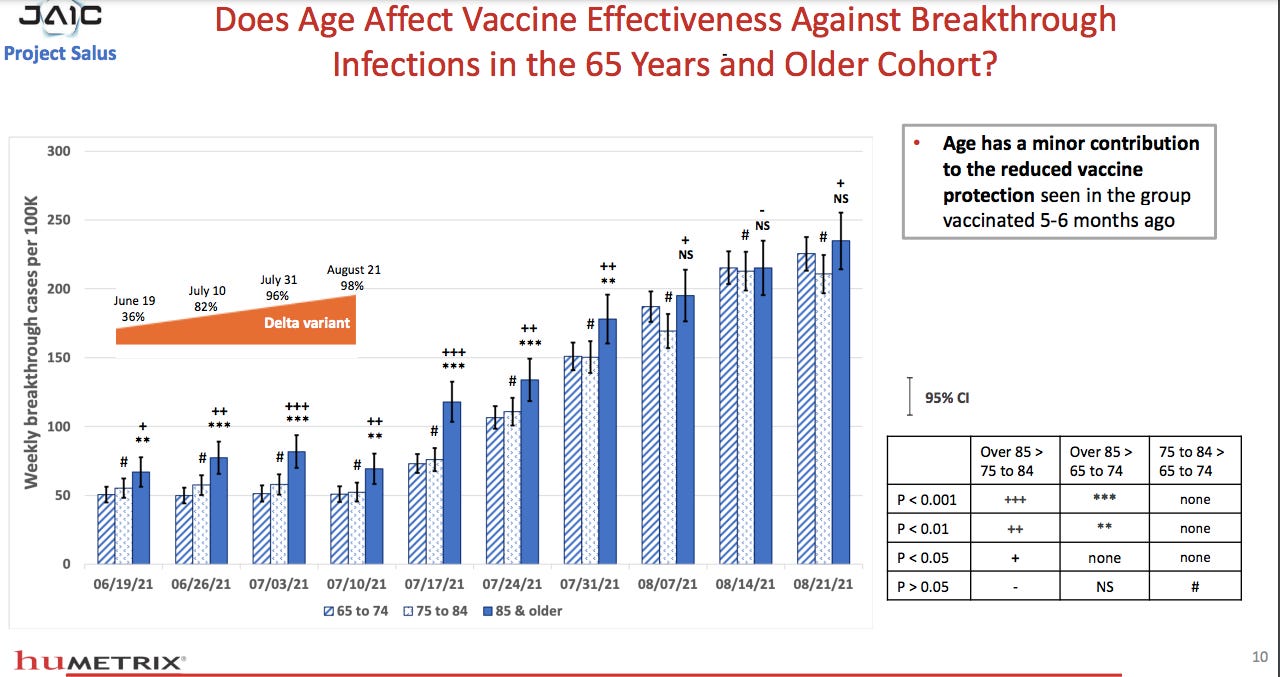
WEEKLY BREAKTHROUGH HOSPITALIZATIONS PER 100K: As time from last vaccination progresses, difference in breakthrough hospitalization rates are greater with increased age. Especially higher for those 85+ vs. 75-84 or 65-74.
Weekly breakthrough CASES per 100K by age: Reduced vaccine protection against infection only minorly associated with age at 5-6 months post-vax. Suggests that acquiring infection relates to vaccines’ lack of ability to prevent infection, NOT age.
Approaching zero vaccine effectiveness against infection by Aug. 7, 2021. Only 62% estimated effectiveness against hospitalization. Where was this notification on CNN or by the CDC or the White House?
This is assessed at the week after delta became >90% dominant. From the first graph, it’s apparent that one week later the effectiveness against hospitalization is even worse, though they say incomplete data due to lag in claims processing. Imagine if they carried this out beyond the beginning of August… And remember, the FDA approval timing with respect to this knowledge…
Here’s a pretty damning slide from a CDC presentation titled “Improving communications around vaccine breakthrough and vaccine effectiveness” July 29, 2021,17 that reads like a PR proposal as to why the above data was hidden from public eye:
Yet in that presentation was this:
** CDC Vaccine Effectiveness Screening Method
Plotted as so:
Furthermore, they conclude the presentation with
“Next steps for CDC:
Communications
– Acknowledge the war has changed
– Improve public’s understanding of breakthrough infections
– Improve communications around individual risk among vaccinated
• Risk of severe disease or death reduced 10-fold or greater in vaccinated
• Risk of infection reduced 3-fold in vaccinated”
However, a few slides earlier concerning “Communications challenges around VE and differential risk,” they specifically state:
“VE estimates represent an average for a group, rather than individual risk
– Risk modified by age, immunocompromising conditions, etc.
– Need to clarify messages around individual protection”
which directly contradicts the above assertion of risk as having x-fold differences for individual risk. They admit individual risk is related to age, other conditions, etc. but then slap on the blanket generalization that individual risk is x-fold different for vaccinated… This is public relations spin-crafting behind the scenes.
So the claim ALL SUMMER INTO FALL heralding a “pandemic of the unvaccinated,” that 99% of those hospitalized (and dying) in the U.S. were unvaccinated was a blatant lie. Per the graph “Total and Breakthrough Hospitalizations in the 65 & Older Cohort,” significant numbers of fully vaccinated individuals were hospitalized even in April (~33%), with numbers near 50% in May and June, only increasing as time went on. This was BEFORE delta neared predominance. Let me repeat: They were flat lying.
Lies such as these have consequences on behavior. And consequences of these behavior differences impact case transmission, case numbers, hospitalizations, and deaths. The flagrantly inaccurate messaging did no public service as far as health is concerned. The exact opposite I would argue.
Protect the vaccine at expense of public health or any other consequence. That’s the message I heard.
More examples of what they weren’t telling the public- in Maryland as of Oct. 18, 2021, “in the last 6-8 weeks, more than 40 percent of people who died in Maryland were fully vaccinated,”18 according to former Centers for Disease Control and Prevention Director Robert Redfield; 76% of deaths from COVID-19 in September in Vermont were fully vaccinated.19
Not to mention that in Spring of 2021 the CDC stopped keeping track of the numbers of breakthrough cases where hospitalization and death did not occur. Say whaaaat? That sure sounds like artificially skewing the numbers to make cases appear to be mostly in the unvaccinated. Per the CDC’s MMWR20, as of May 1, 2021 they stopped “monitoring all reported COVID-19 vaccine breakthrough infections” to investigate “only those among patients who are hospitalized or die, thereby focusing on the cases of highest clinical and public health significance.” This is the organization the U.S. public is trusting with regard to COVID?! I have never seen a cooking of the books be so flagrant.
With all due respect CDC, it is highly significant to public health if there are increased numbers of breakthrough cases that aren’t severe since this:
1) further indicates waning vaccine effectiveness/vaccine failure;
2) significantly affects transmission to others (unvaccinated, partially vaccinated, or fully vaccinated), um propagating the pandemic?!;
3) affects transmission to those who may be even more vulnerable (regardless of vaccination status);
4) affects behavior of those who are vaccinated, possibly leading them to unknowingly spread infection because this risk was hidden;
5) further muddies the water regarding the rationale behind vaccine mandates.
Sweeping data under the rug is not public health or science.
Further regarding decreased vaccine effectiveness, yes, waning immunity is an issue, but this was always going to occur to some degree, especially when only considering antibody levels. The human immune system does not keep antibody levels exceedingly high indefinitely. It would be wasteful and potentially harmful. The way that prior immunity helps is to allow for the robust response quicker, which can better curb the infection and sometimes even prevent its establishment. It goes against our body’s natural immune process to artificially maintain antibodies at high levels. Apart from the potentially harmful spike proteins themselves,21 22 23 spike-specific antibodies for SARS-CoV have been implicated in the immunopathologic response causing severe acute lung injury24 25 and for SARS-CoV-2 have been shown to have affinity for many different tissues in our body.26 27 28 29 This seems to be begging for autoimmune damage, short-lived or otherwise. Indeed, a 2019 review of previous CoV vaccine development acknowledged that “since severe CoV disease maintains an immunopathogenic component, a successful vaccine must strike a balance between protection and excessive immune activation.”30 Depending on the tissue, this type of damage can cause scarring, which can permanently affect future function.
Since these safety signals (among many other types) may not be readily apparent in the short-term, mass vaccination and now boosters are not a cautious approach to testing these vaccines. Especially for those who are at low risk of severe COVID. In fact, the Vaccines and Related Biological Products Advisory Committee (VRBPAC) OCTOBER 22, 2020 presentation(14) released an extensive list of possible adverse events, with autoimmunity underlying a great proportion of them.
SLIDE 16:
Nearly all of these have been reported in VAERS and in drastically higher numbers than the combined totals of all other previous vaccines’ adverse events in the ~30-year history of the reporting system. The number of adverse events that triggered the suspension of previously failed vaccine programs (such as 3 deaths during 1976 swine flu vaccine program; or dozens of Guillain-Barré syndrome cases from the same vaccine program a few months later31 32; or 18 hospitalizations with 2 deaths for RSV33(24)) was orders of magnitude lower than any already tallied now for these COVID-19 vaccines.
I would wager that nearly everyone who got the vaccine in early 2021 was never informed of these possible risks either. That the vaccine could actually make a future infection worse than if they hadn’t been vaccinated (“Vaccine enhanced disease” from above). Or told that even if they were vaccinated they can still get COVID, still be hospitalized from COVID, and still die from COVID. Informed consent was beyond breached.
Additionally the “experts” would have been familiar with the concept of immunosenescence- that as individuals age, there is a “progressive reduction in the ability to trigger effective antibody and cellular responses against infections and vaccinations.”34 As mentioned, previous studies using older mice for SARS-CoV vaccine development demonstrated this. In their COVID-19 vaccine clinical trials the >65 yo demographic was only approximately 25% of the treatment and placebo arms (so only ~12.5% at most actually got the vaccines per trial), with >75 yo greatly underrepresented, likely not by accident (as this would have undoubtedly sullied their numbers, see previous Part 6). Remember, it was known that these were the exact demographic who most require protection. How does it make sense to not test the proposed treatment with enough subjects for whom the treatment effect is most needed? Let alone not structure the trials to test whether the vaccines actually have a statistically significant reduction in hospitalization or death! (Thus is the problem with determining statistical significance for a disease with such a low rate of severity in the general population to begin with.)
Additionally, immunosenescence has been a well-established problem for influenza vaccines.35 36 And maybe more interesting, antibody titers due to flu shots in the elderly have been shown not to correlate well with protection (instead T cell responses may be more predictive).37 38 Remember, antibodies have been the selling point of the COVID vaccines. Yet these known limitations somehow weren’t applied when considering the leading protective strategy for the most vulnerable populations? This also relates to LIE 7 (see below).
Bottom line: The COVID-19 vaccines only confer partial immunity, which is in direct opposition to Requirement #3. Herd immunity cannot operate. They knew in advance that this was the overwhelming likelihood and then covered it up when the vaccines weren’t performing as initially promised.
Sure seems to fit this statement from The Brighton Collaboration VAED working group:
“In the event of low/poor vaccine efficacy, infection will occur in vaccinated subjects, with breakthrough disease associated with viral replication.”39
But it would have been bad for business to admit it up front. Knowing beforehand that they would need to rely on the post-authorization data (not only in terms of increased numbers but also length of time) to determine:
1) any benefits in reduction of hospitalizations, deaths, transmission prevention;
2) durability of immune response and correlates of protection;
3) cross-immunity to new variants;
4) vaccine enhanced disease;
5) immune response in special populations;
6) special-interest adverse events; &
7) long term safety,
they certainly had a conflict of interest when it came to generating volunteers to participate. It could be argued that they said what people wanted to hear after being made fearful and desperate for months in order to accomplish this goal.
LIE 7: Boosters likely wouldn’t be necessary.
When selling the idea of these vaccines, they insinuated that the protocol would be a two-shot (or one-shot) series and boom, you’re done. If asked about boosters in summer and early fall, they might say something like, “We hope they won’t be necessary.” Yet every piece of evidence based on previous research40 or established scientific principles was screaming that these will undoubtably be the protocol. “Many efforts have been directed to develop vaccines against human CoV infections in recent decades, but a limiting factor is the degree of cross-protection rendered by these vaccines due to their extensive sequence diversity.”41
(More support from many prior paragraphs and cited studies Parts 1-6).
In fact, Pfizer and Moderna had been developing and testing updated vaccines based on other strains, as early as January 2021 (announcement by Moderna for the South African variant).42 Additional clinical trials for boosters began May 2021.43 Hmm, again, sure sounds like they knew more behind the scenes (this would not be “one and done” or “two and done”) than what they were admitting to the public.
Plus, with previous vaccine development and COVID-19 vaccine clinical trials data, they knew that boosters would be required to combat the very nature of the virus and the short-lived, imperfect immunity generated, especially if we chose to rely on vaccine-induced immunity to address the pandemic. Just as is the case for annual flu shots. Logic would infer that boosters would be the only artificial way to achieve the possibility of longer protection from the vaccine route.
That is if this strategy even should be employed in the midst of a pandemic with the vaccine characteristics as they are and the virus properties as they are. Given selective evolutionary pressure generated by the spike protein vaccines, mass boosting will only serve to strengthen this selective pressure. And possibly at the risk of increased adverse events both to the individual and world population at large.
Here’s but one possible worse-case outcome.
A nightmare scenario arose in the poultry farming industry from a mass vaccination situation. The vaccine for Marek’s disease in chickens is a leaky vaccine, only preventing severe disease while allowing infection and transmission.44 45 This has allowed for an aberration of the natural selection process that would normally occur if a highly lethal variant were to arise; instead of the variant rapidly fizzling out due to the high, rapid fatality, the vaccine allows for the evolution and propagation of highly lethal variants. One paper suggested that “the risk of outbreaks of hyperpathogenic strains be considered wherever disease interventions improve host survival without preventing pathogen transmission.”46 Given that the occurrence of hyper virulent variants attaining increased success in transmission from the widespread use of a leaky vaccine is not limited to only chickens (occurs in other animals with different infectious agents (46)), it seems the lack of caution bordering on irresponsibility may not be the best approach for a pandemic and vaccine with these circumstances.
This doesn’t even elaborate on other concerning topics
(Call this my Good Will Hunting moment):
Hmm, so why shouldn’t we ignore other concerning topics? It’s a tough one, but I’ll take a shot
1) vaccine-associated disease enhancement (VADE)/ vaccine-associated enhanced respiratory disease (VAERD), referring to both antibody-dependent (ADE) and TH2 cell-dependent disease exacerbation (Fauci even admitted in March 2020 in a press briefing (~minute 3) that we’ll be looking for these to be “interspersed” with the vaccine rollout);
they have a special committee (The Brighton Collaboration VAED working group formed in March 2020) giving guidance on this, though they admit it will be very challenging to determine, even more so if there are no placebo groups (39) (the vaccine manufacturers eliminated them after 2 months);
2) vaccine enhanced disease in animal models-
SARS and MERS vaccine development showed this repeatedly in animal testing;
Chlamydia trachomatis in monkeys47, influenza in pigs and ferrets48 (57,58), Mycoplasma pneumoniae in mice49 and Mycoplasma spp. in cows50;
3) other vaccines previously approved or in human trials with VADE, some were suspended
2009 H1N1 flu, RSV, Denge virus, measles virus, Mycoplamsa pneumoniae, HIV, and rotavirus to name just a few examples51 52 53; many of these effects were only discovered years after use to boot! Yet we rolled these COVID vaccines out after 2 months and to billions of people…
We still too often don’t know what causes it:
“The difference between these observations in human and mice could be attributed to several factors including: difference in antibody types (classes) between the two species, titer of antibodies in the blood, the challenge model used in mice, and many others.”
specifically antibody dependent enhancement (ADE) demonstrated either with vaccines or using in vitro cell replication studies: influenza, West Nile Virus, Denge virus, RSV, Zika virus, Chikungunya virus, Ebola virus, Japanese Encephalitis Virus, Epstein-Barr virus, herpes simplex virus, foot-and-mouth disease virus, measles virus, rabies virus, human metapneumoviruses.54 55 56 57 58
taken together, VADE is NOT an uncommon problem in vaccine R & D; ignoring prior cautionary advice was risky in the most favorable of lights
4) prior failed pandemic vaccines due to serious adverse events (1976 swine flu, 2009 H1N1 flu);
2009 H1N1 flu vaccine (Pandemrix) resulted in >1,300 children diagnosed with narcolepsy; discovered 1-2 years later, but legal matters took even longer:
“Eight years after the <2009> pandemic influenza outbreak, a lawsuit alleging that GlaxoSmithKline’s Pandemrix vaccine caused narcolepsy has unearthed internal reports suggesting problems with the vaccine’s safety…In October 2009, the US National Institutes of Health infectious diseases chief, Anthony Fauci, appeared on YouTube to reassure Americans about the safety of the “swine flu” vaccine:
“The track record for serious adverse events is very good. It’s very, very, very rare that you ever see anything that’s associated with the vaccine that’s a serious event.”59
So glad we have the same guy doing the same thing again…;
For the 1976 swine flu:
This compilation highlights both:
5) serious adverse events causing suspension of other approved vaccines:
“The Cutter incident” in 1954-55 (when the inactivated polio vaccine was just developed and approved) where 200,000 children were accidentally injected with a formulation of poliovirus vaccine in which the virus was not adequately killed, leading to 40,000 infections (from injection and community spread), 200 paralyzed and 10 deaths.60 61
live-attenuated oral poliovirus vaccine (OPV; Sabin) reverting to virulent form and causing vaccine-associated paralytic poliomyelitis (VAPP) and vaccine induced community outbreaks (called circulating vaccine-derived poliovirus or cVDPV), resulting in ~ 6-8 cases of paralysis each year in the U.S. in the 1980s & 1990s; this led to OPV discontinuation in 2000 (replaced with a modified Salk inactivated vaccine- IPV)(59) 62 63 64
LYMErix (vaccine against Lyme disease) withdrawn from market amidst ongoing litigation and negative publicity for safety concerns (arthritis) resulting in failing market sales 65; the negative media coverage and public reaction to it tanked this product…
They learned very well to not let this happen again. Legal immunity to protect profits wasn’t enough; can’t be immune to media coverage unless you control that coverage…
6) A skewed proportion of adverse events concentrated in very few lot numbers for COVID-19 vaccines compared to a low-level distribution of adverse events across nearly all lot numbers for all flu vaccines (from VAERS data anaylsis) suggestive of dose range-finding or dose-ranging study;66
7)original antigenic sin (OAS), a principle in virology/immunology whereby an initial encounter with a new pathogen primes the immune system to develop a skewed response to future infections with the same or similar pathogens;67 68 69
8) repeated annual flu shots can negatively affect immune response to new flu virus challenge (termed negative interference);70
9)the story of myxoma virus in rabbits as a cautionary tale of virology and evolution—
the virus was released intentionally into Australia and France in the 1950s as a means of pest control; however, rabbit evolution, likely paired with viral attenuation, quickly overcame the once highly lethal virus. So rabbit populations recovered. But the evolutionary fight didn’t stop there. Myxoma virus continued its evolution to become more virulent once again, thought to result from genetic resistance of some rabbits allowing for prolonged infection (similar to leaky vaccine problem).71 (46)
10) concept of high-zone tolerance:
“Immunologic tolerance is a natural phenomenon defined as a non-responsivity state to an antigen which is triggered by a previous exposition to it. The same antigen material can behave as a tolerogen or an immunogen, depending on how this antigen is presented to lymphocytes. Currently, it is well known that the injection of large quantities of soluble non-aggregated antigens, in the absence of adjuvant, generates what is known as High Zone Tolerance, a failure in obtaining a specific T-cell response. This phenomenon culminates an anergy of the immune system, in which tolerogen-specific B cells are eliminated.” 72
“Classical tolerance is dependent on the dose and the timing of antigen delivery, with “low-zone” tolerance referring to low antigen doses over prolonged periods of time and “high-zone” tolerance dealing with high doses of antigen overwhelming the immune system [88]. In both cases, antigens are specifically recognised and induce central or peripheral deletion of reactive T- and B-cells, while at the same time generating antigen-specific Tregs (called Tr1 cells).73
could this be applicable regarding these vaccines and boosters? seems like a valid concern;
11) VAERS and EU adverse events surveillance data for COVID-19 vaccines compared to all others in the past several decades;
12) commonly used approved drugs pulled from market or from certain uses (Vioxx, thalidomide, Phen-Phen, diethylstilbestrol (DES), etc.); Additionally:
Nearly 1/3 of FDA approved drugs from 2001-2010 had problems74:
“Seventy-one of the 222 drugs approved in the first decade of the millennium were withdrawn, required a "black box" warning on side effects or warranted a safety announcement about new risks…
“…the FDA approves drugs faster than its counterpart agency in Europe does and that the majority of pivotal trials in drug approvals involved fewer than 1,000 patients and lasted six months or less.
It took a median of 4.2 years after the drugs were approved for these safety concerns to come to light, the study found, and issues were more common among psychiatric drugs, biologic drugs, drugs that were granted "accelerated approval" and drugs that were approved near the regulatory deadline for approval.”75
13) autoimmune diseases possibly linked to prior vaccines or infections— Hepatitis B and multiple sclerosis78; H1N1 vaccine and narcolepsy79, HPV and systemic lupus erythematosus (SLE)80; LYMErix and arthritis (64); enteroviruses, especially group B coxsackievirus and Type I diabetes mellitus;81
14) The Vaccine Act 1986 and PREP Act 2005 (EUA designated products) protecting vaccine manufacturers from liability;82
“In 2005, Congress passed a tort shield law, the PREP Act, to protect manufacturers of drugs and other “covered countermeasure[s],” including vaccines, from the risk of damages in the event of a declared public health emergency. This statute goes considerably further than the Vaccine Act to create an exclusive limited administrative remedy. The PREP Act disallows those injured to apply to the NVICP; they must apply to an administrative program that HHS administers itself. The PREP Act covers vaccines, antidotes, medications, medical devices, and other products used to respond to pandemics and biological and chemical threats. If the HHS Secretary declares a public health emergency, then liability protection covers not only manufacturers, but all medical administrators of the covered countermeasures to prevent, treat or mitigate an epidemic. The Secretary’s declaration is not reviewable by any court. The PREP Act sets up an administrative Countermeasures Injury Compensation Program (CICP) in HHS for people seriously injured from the use of products under a PREP Act declaration. The CICP has a one-year statute of limitations. While a claimant may hire a lawyer, unlike in the NVICP, the CICP does not pay any attorney fees. The CICP offers no hearings or appeals from the CICP decisions; however, a claimant may request reconsideration of her claim within sixty days if CICP rejected it on the first review. There are no published records of CICP’s compensation decisions, so it is impossible to analyze them. CICP’s website lists medical expenses, lost employment income, and survivor death benefits as possible compensation, but it is unclear whether or to what extent CICP has paid them, as there are no published decisions... With effective access only to an administrative tribunal, with a one-year statute of limitations, and with no opportunity for appeal or review in any court, consumers have exceptionally limited recourse under the PREP Act. Most consumers in the United States pay little attention to the fine print when they get vaccines. Yet whether a person receives a seasonal flu vaccine or an emergency pandemic one, such as the H1N1 flu shot, could make a world of difference in what recourse might be available in the event of injury…
…some have suggested that the PREP Act incentivizes manufacturers of emergency medical products to willfully disregard or consciously avoid problematic risk information so that they cannot fall within the Act’s “knowing” requirements. In an article in the Journal of the American Medical Association about the role of litigation in defining drug risks, the authors cite a memorandum from a drug company executive, which states: “If the FDA asks for bad news, we have to give, but if we don’t have it, we can’t give it to them.” It appears that the PREP Act may incentivize precisely this kind of thinking.”
From the same 2018 paper, also highly relevant to motive/mindset:
“In January 2017, a number of international public and private actors formed a new global institution, the CEPI, to help create vaccines for emerging epidemic threats, particularly in the developing world. The governments of Germany, Japan, and Norway, together with the Bill & Melinda Gates Foundation and the Wellcome Trust, made initial investments of $540 million; the European Commission has pledged 250 million euros. India is expected to donate as well. Several vaccine manufacturers, including GlaxoSmithKine, Johnson & Johnson, Merck, Pfizer, Sanofi Pasteur, and Takeda, became CEPI partners rather than donors, as did the intergovernmental WHO and non-profit Doctors Without Borders. CEPI’s partners announced this new effort at the World Economic Forum in Davos, Switzerland in early 2016…
Although vaccines against Ebola were developed, they were available only after the epidemic was waning and many thousands had already died. CEPI aims to create a new infrastructure that will ensure greater global preparedness for future pandemics by developing and stockpiling the vaccines most likely to be useful. The first three vaccines CEPI plans to develop are for MERS, Lassa, and Nipah viruses, which are on the WHO’s list of priority pathogens. These infectious diseases primarily affect the developing world, and the marketplace on its own likely would not develop vaccines against these diseases quickly. CEPI takes a comprehensive approach, ensuring support for vaccine candidates through late preclinical studies and will support vaccine technologies that enable rapid development. CEPI’s scientific advisory committee chose these three diseases based on potential for public health impact, risk of outbreak, and feasibility of vaccine development. CEPI also plans to sponsor research into a new class of vaccines, known as RNA vaccines, to allow much faster development.*
* “The beauty of these RNA vaccines is that once you develop a way of delivering the RNA into a patient—some kind of scaffold or shell—you can theoretically customize it to deal with all kinds of diseases. Just swap the Ebola RNA for that of some other virus, and you’d have a new vaccine in a matter of weeks. And if you could show that the scaffold is safe, no matter whose RNA it carries, then you could speed that new vaccine through the regulatory process.”
CEPI also notes that it will collaborate with WHO objectives, including “[d]evelopment and implementation of new norms and standards adapted to and appropriate for an epidemic context.” While this statement is vague, it appears from news reports that the CEPI board is exploring different models for manufacturer liability protection and victim compensation. A PowerPoint on the WHO website notes that CEPI must create “market security,” comprised of “positive externalities,” “minimal disruptions,” and “market predictability.” Furthermore, CEPI declares that it “will rely on WHO as the global normative lead agency on health.”
Dr. Jeremy Farrar, director of Wellcome Trust and a CEPI board member, said to the New York Times that CEPI favors the U.S. NVICP model, under which vaccine makers cannot be sued directly, but must contribute money they collect as excise taxes from consumers to a fund that compensates those injured. But Andrew P. Witty, chairman of GlaxoSmithKline, said that industry preferred protection along the lines of the PREP Act, “which exempts vaccine makers from all liability—except for willful misconduct—once the [Secretary of HHS has] declare[d] a public health emergency.”
This sure seems to hint at what is meant by “create a new infrastructure” and “must create “market security,” comprised of “positive externalities,” “minimal disruptions,” and “market predictability”and “[d]evelopment and implementation of new norms and standards”:
15) prior big pharma lawsuits for fraudulent marketing, examples:
in 2009 “Pfizer to Pay $2.3 Billion for Fraudulent Marketing” plus an additional $14.5 million in 2011 to resolve False Claims Act allegations related to its marketing of the drug Detrol;83 84
“Pfizer Settles a Drug Marketing Case for $491 Million” in 2013;85
Janssen subsidiary of “Johnson & Johnson to Pay More Than $2.2 Billion to Resolve Criminal and Civil Investigations” in 2013 (a decade after it perpetrated these acts;86
“Pharmaceutical Giant AstraZeneca to Pay $520 Million for Off-label Drug Marketing” in 2010.87
16) ignoring and censoring early treatment, conveniently putting all of our eggs in the vaccine basket (the existence of an effective therapeutic could threaten the vaccine EUA, possibly preventing it altogether, or possibly incur less voluntary uptake of the vaccines). In defense of their application for EUA December 2020, Moderna states:
“2.4 Alternatives for Prevention of COVID-19
No vaccine or other medical product is FDA approved for prevention of COVID-19. On December 11, 2020, FDA issued an EUA for the Pfizer- BioNTech COVID-19 vaccine for active immunization for prevention of COVID-19 due to SARS-CoV-2 in individuals 16 years of age and older. However, the Pfizer-BioNTech COVID-19 vaccine is not an approved product, and furthermore is not available in quantity sufficient to vaccinate all persons in the U.S. for whom the vaccine is authorized for use. On October 22, 2020, FDA approved remdesivir for use in adult and pediatric patients 12 years of age and older and weighing at least 40 kilograms for the treatment of COVID-19 requiring hospitalization. Several other therapies are currently available under emergency use authorization, but not FDA approved, for treatment of COVID-19. Thus, there is currently no adequate, approved, and available alternative for prevention of COVID-19.” (emphasis added);88
By definition there are four requirements for an EUA, part “d. No alternative” states:
“For FDA to issue an EUA, there must be no adequate, approved, and available alternative to the candidate product for diagnosing, preventing, or treating the disease or condition.”89
17) choosing Remdesivir as the only approved drug for months and only once disease was severe, despite it being pulled from its only previous human trial due to inferior results as compared to 3 monoclonal antibody treatments90; Remdesivir had the highest all cause mortality rate at 53% (93/175) and performed worst for decreasing viral clearance. (READ- why were the more successful monoclonal antibody treatments NOT broadly used from the start instead of Remdesivir?);
Fauci even said in an interview in 2019 regarding this trial:
“It means that we do have now what looks like treatments for a disease which not too long ago we really had no therapeutic approach at all… And the earlier the better, as in any disease.”91 (emphasis added)
Indeed, another conclusion from the trial was that a longer duration of symptoms before treatment was associated with worse outcomes…
yet Remdesivir became THE DRUG for COVID treatment with a protocol that waited until patients required hospitalization (precisely prescribing a longer duration of symptoms before treatment!);
not to mention that the World Health Organization recommended against using Remdesivir throughout 2020 to present92; something doesn’t add up;
monoclonal and polyclonal Abs were extensively studied for SARS and MERS and shown to be effective93:
Consequently they were “the leading proposed treatment”94 for MERS
“administration of neutralizing MAbs specific to the SARS-CoV spike protein may be a useful strategy in post-exposure treatment and prophylaxis in at risk populations.” => they could have used these from the beginning
18) deep conflicts of interest among government officials, bureaucratic agencies, main stream media, and big pharma;
19) financial incentives for the health systems:
to admit COVID positive patients-
HHS paid $76,975 per COVID admission during the first round and $50,000 per COVID admission during the second (with additional rounds added later, Phase 4 as of 9/29/21, still ongoing95) if between Jan. 1 and June 10, the hospital had either 161 COVID admissions, at least one COVID admission per day, or averaged higher than the national average ratio for COVID admissions/bed.96 Phase 1 & 2 provided relief for 83% of total COVID admissions during this time frame;
These “High-Impact Area Targeted Distributions” are separate from “General Distribution” and other assistance available (other targeted distribution types, FEMA, etc.), which are not mutually exlcusive;
hospitals have 12-18 months to utilize these funds for “allowable expenses or revenue losses attributable to coronavirus,”97 which has a very generous definition (see below); in this way, the moneys disbursed are like COVID loans or grants with strings attached- there are Terms & Conditions;
interesting qualifying expenses include: hiring bonuses and retention payments; paying multiple employee salaries with a cap of $197,300 per employee (ex. if 25 RNs, their $1.25 million in total salaries fall below this cap!); benefits; paying taxes; human resources; costs not reimbursed by another source or in excess of that reimbursement; food services; laundry;
maintaining supporting documentation for qualifying costs is required to keep this money, almost none has been returned;
good thing “HHS broadly views every patient as a possible case of COVID-19” and providers only need to have provided service to “possible or actual cases of COVID-19” after Jan. 31, 2020.
but to be eligible they need to not be terminated from Medicare, be excluded from participation in Medicare or Medicaid, and not have Medicare billing privileges revoked; makes me wonder how this could come into play for the healthcare vaccine mandates…
shutting down elective procedures (like surgeries), among other COVID circumstances, undercut a main source of hospital revenue, thereby putting hospitals in a position early in 2020 (and beyond) to need or desire financial assistance from the HHS funding which then requires justification to keep this money, such as COVID + patients, COVID hospitalizations, COVID patients on ventilators, and COVID deaths. Taking the money up front incentivizes generating qualifying expenses.
inflated reimbursements from CMS and private insurance companies for COVID-19:
Medicare patients discharged with a primary or secondary diagnosis of COVID-19 are reimbursed with a 20% bump above the usual Diagnosis-Related Group (DRG) under section 3710 of the CARES Act; example $13,155 for inpatient admission; $39,896 for ventilator >96 hrs.;98 99
In 2017 for similar treatments mentioned above, private insurance on average paid $100,000 for ventilator >96 hrs. compared with $40,000 for Medicare and $34,000 for inpatient admission due to respiratory infection vs. $13,000 for Medicare; the trend of 2 times higher payments for private insurance was also found for COVID100; from this study ~1/3 of inpatients had private insurance;
and not deviate from the NIH protocols, even when said protocol is failing:
the PREP Act covers any provider (in addition to manufacturer, administrator, distributor) from liability as long as public health emergency is declared and said providers utilize FDA-ordained “covered countermeasures”101;
reading between the lines: hospitals therefore might have a huge disincentive to allow their practitioners to deviate from NIH protocol; I argue that this is a driving reason for lockstep obedience by hospitals and providers; however, this doesn’t explain the reason for NIH’s severe bottleneck on “approved” protocols especially for early treatment;
with all this “free” money on the line, there’s millions of reasons to play along, go with the flow, toe-the-line; and anyone jeopardizing this is expendable;
20) pushing COVID vaccines that neither stop infection or transmission on healthy children for whom natural immunity would be superior, and in whom the risk of getting severely ill or dying from COVID is statistically almost zero, making the already-known risks and unknown possible risks outweigh their use for younger populations. Many of the previously failed vaccines took 1-2 years or longer for their adverse events to become apparent. Plus:

Link to FDA advisory committee meeting:
skip to ~5:43:16 (yes, 5 HOURS, 43 minutes)
Dr. Eric Rubin, FDA advisory committee member and Editor-in-chief of the New England Journal of Medicine who states:
“We’re never going to learn about how safe this vaccine is unless we start giving it. That’s just the way it goes. That’s how we found out about rare complications of other vaccines like rotavirus vaccine.”
The rotavirus vaccine he refers to (RotaShield™) got pulled only 14 months after licensure due to the serious adverse event called intussusception (a potentially life-threatening intestinal blockage).(65)
21) the concept of natural boosting (occurs every year for multiple circulating viruses), which is hindered by prolonged mass mitigation measures like school closures, reliance on virtual encounters, lockdowns, etc.
as an example regarding influenza, the authors mention that an absence of pathogen transmission “will prevent natural boosting… and could lead to waning immunity;”102
this could apply not only to SARS-CoV-2 but many other commonly circulating infectious viruses;
22) concerns that pharmacovigilance mechanisms are not adequate:
admission by a top scientist in vaccine development, Dr. Martin Howell Friede (coordinator, Initiative for vaccine research (IVR), WHO), that the adverse event surveillance systems for long term effects and systemic adverse events for Phase II/III clinical trials are lacking (from the Global Vaccine Safety Summit- Geneva Switzerland, Dec. 3, 2019)103;
Harvard study showing that these events are drastically underreported in such systems, with “fewer than 1% of vaccine adverse events”104 reported;
23) distribution studies showing that the injected vaccine components do not stay only at the injection site;105 106
a European Medical Agency (EMA) study published February 19, 2021, reported on Pfizer’s Covid-19 mRNA vaccine BNT162b2.
which further referenced PF-07302048_06 Jul 20_072424, a biodistribution study in mice and rats utilizing radiolabeled lipid nanoparticles (LNP) and luciferase modified RNA (to track their distribution after injection):
radioactivity was “detected in most tissues, with the greatest levels in plasma observed 1-4 hours post-dose”
“Over 48 hours, distribution was mainly observed to liver, adrenal glands, spleen and ovaries, with maximum concentrations observed at 8-48 hours post-dose.”
that these surrogate markers were found beyond the injection site, strongly indicates that lipid nanoparticles and the subsequently manufactured spike proteins can be distributed throughout the body (remember them assuring “it only stays in the arm”?)
24) FDA approval of Pfizer’s vaccine relied on efficacy data stopping in Spring of 2021, rather than including what was available up until its request for approval (into August);
furthermore a study available mere days after the approval concluded vaccine efficacy had waned to 47% after 5 months (and these authors had extensive affiliation with and funding by Pfizer);
50% is the threshold of efficacy to remain under EUA, with the lowest bound in the confidence interval at >30%;
running the numbers this way allows for the greatest likelihood to calculate higher VE;
25)the “approved” Comirnaty is not and has not been available in the U.S., yet the healthcare provider fact sheet states that the two products are “legally distinct”. Plus the “approval” age is >16 years of age. And doesn’t cover 12-15 yo, 5-11 yo, boosters of any age group. At the exact same time, the EUA for Pfizer-BioNTech vaccine was extended concerning these other purposes (12-15, 5-11, boosters of any age group).107 This remaining EUA distinction makes everyone exempt from liability in the event of injury or adverse effects (see previous regarding PREP Act).
26) THESE VACCINES REMAIN EXPERIMENTAL. Study conclusion dates from the original clinical trials extend into 2023, with additional focused study conclusion dates into 2024 and 2025.108 Reminder that their EUA documents (December 2020) admit nearly every important criterion requires further study once the EUA is permitted, making guinea pigs out of any who volunteered but without informed consent that this would be the case;
27) Moderna’s history of never before having a successful product come to market; a 2017 article109 with employee interviews saying that previous mRNA-based treatment failures due to safety problems (incurred from repeated dosing! Concerns for these new frequent boosting protocols?!) would mean they will have to undertake the less-profitable vaccine development route:
“Unable to press forward with that technology, Moderna has had to focus instead on developing a handful of vaccines, turning to a less lucrative field that might not justify the company’s nearly $5 billion valuation.
“It’s all vaccines right now, and vaccines are a loss-leader,” said one former Moderna manager. “Moderna right now is a multibillion-dollar vaccines company, and I don’t see how that holds up.” (emphasis added)
COVID to the rescue. How coincidental that COVID-19 events would transpire to resurrect their doomed industry with partnership from NIAID of the NIH and CEPI to the tune of billions;
Additionally with the EUA swooping in to lower the standards for releasing these products on a grand scale. Per “Emergency Use Authorization of Medical Products and Related Authorities” official FDA document January 2017
“Medical products that may be considered for an EUA are those that "may be effective" to prevent, diagnose, or treat serious or life-threatening diseases or conditions…The "may be effective" standard for EUAs provides for a lower level of evidence than the "effectiveness" standard that FDA uses for product approvals.”110
the partnership between Moderna and NIH for the COVID-19 vaccine:
“CEPI has announced funding to Moderna for comparing mRNA therapeutics and vaccines. They will design and manufacture an mRNA vaccine in collaboration with the Vaccine Research Center (VRC) of the National Institutes of Allergy and Infectious Diseases (NIAID), a part of the National Institutes of Health (NIH).”(40);
conflict of interest for NIH to be promoting these; maybe helps explain the vaccine tunnel vision by Fauci, Collins, CDC, FDA, etc.
now open the flood gates for new vaccine human clinical trials using mRNA just in the past year;
additionally a 2016 article in Nature accused Theranos and Moderna of producing “research not fit to print”111
Hmm, what’s become of Theranos recently, once previously touted as an innovative biotech company?112
28) core principles of medical ethics (such as autonomy) and informed consent;
29) advocation for human infection challenge studies (HCS; intentionally infecting volunteers with a test pathogen under testing circumstances, for example after vaccination) for COVID-19 early in the pandemic;113 raises suspicion about just what we are involved in considering the lack of true informed consent and false messaging
regarding SARS-CoV vaccine research- “Human vaccine effectiveness studies will only be possible in the context of SARS re-emergence.”114
The practice has a checkered and extensive history, even as recent as the 20th century;
A perplexing discordance:
Recent rare cases of serious harms, such as possible myocarditis or MI among malaria challenge study participants in their 20s post-vaccination, led to the exclusion of those with cardiac risk factors from future HCS. The precaution was taken after only 2 cited case reports (2009 and 2014) from two different trials (2009 and 2014) after these studies have been conducted for decades.
Similarly an influenza human challenge study investigating the use of peramivir as a prophylactic agent and one case of myocarditis in a 21-year-old, previously healthy individual with no prior cardiac history. “Despite the subject's return to baseline health, and despite the lack of direct causality linking myocarditis to the influenza challenge stock, no further influenza challenge studies were conducted in the US for nearly a decade later.”(35)
Sure seems like they took myocarditis seriously for these studies…where’s that same concern regarding the COVID vaccines??…I wonder whether the liability protection in such studies differs from PREP Act vaccine liability protection and could have something to do with it…
For human infection challenge studies the question is how much is too much compensation (and therefore construed as coercive) or how little is too little to offer for participation.115 Interesting that we’ve seen a slew of carrots (free donuts, french fries, hamburgers, gift cards, entry into lotteries, etc.) before being given the stick (lose your job, incur fines, barred from portions of society).
30)long historical track record of unethical medical procedures and experimentation on human populations sanctioned by government, doctors, etc.116 117:
slaves in Southern U.S., forced sterilization (Buck v Bell 1927) of 70,000 “unfit” individuals; Tuskegee study; Nazis in Germany, etc.
31) parallels in 20th century world history with respect to the rise, establishment, and maintenance of totalitarian regimes; Fascism in Italy, Hitler & Nazis in Germany, socialism in Soviet Union;
https://www.pbs.org/video/the-story-of-fascism-in-europe-pthanf/
32) aims to address “vaccine hesitancy” and “misinformation” prior to beginning of COVID pandemic:
Trusted News Initiative (initially European Broadcasting Union (EBU), Facebook, Financial Times, First Draft, Google, The Hindu, and The Wall Street Journal) convened July 2019 to address “the rise of misinformation and bias”:
partners currently within the TNI are: AP, AFP; BBC, CBC/Radio-Canada, European Broadcasting Union (EBU),Facebook, Financial Times, First Draft, Google/YouTube, The Hindu, Microsoft , Reuters, Reuters Institute for the Study of Journalism, Twitter, The Washington Post.,118
Event 201 in October 2019- pandemic simulation and planning exercises at Johns Hopkins,119
discussions from Global Vaccine Safety Summit- Geneva Switzerland, Dec. 3, 2019 about vaccine messaging problems and paucity of safety data
European Commission Roadmap on Vaccination Q3 2019120:
“guidance on countering vaccine hesitancy,” “combating online vaccine misinformation” through the “Commission Communication on tackling disinformation” since 2018;
“Consider investing in behavioural and social science research on the determinants of vaccine hesitancy across different subgroups of the population and healthcare workers”
plan for “a common vaccination card/passport for EU citizens” with the “feasibility study” coincidentally projected from 2019 to 2022…
33) deliberate disinformation capitalizing on the first impression rule- flashy story/headline one day, correction/retraction to be seen by no one days later:
“An article on Thursday about recommended single doses of the coronavirus vaccine for children in some countries described incorrectly the actions taken by regulators in Sweden and Denmark. They have halted use of the Moderna vaccine in children; they have not begun offering single doses. The article also misstated the number of Covid hospitalizations in U.S. children. It is more than 63,000 from August 2020 to October 2021, not 900,000 since the beginning of the pandemic. In addition, the article misstated the timing of an F.D.A. meeting on authorization of the Pfizer-BioNTech vaccine for children. It is later this month, not next week.”121
Ivermectin fake story from The Rolling Stone, “Gunshot Victims Left Waiting as Horse Dewormer Overdoses Overwhelm Oklahoma Hospitals, Doctor Says.” Except the quoted doctor hadn’t worked in the ER for over two months. The hospital’s statement further mentioned that no ivermectin-related cases had been seen, including no overdoses, and no patients have been turned away. Even the photograph used with the story was false122 123 124
Precisely emulates the disinformation campaign done by Big Tobacco, media, and the U.S. government for decades in order to intentionally promote confusion and doubt in the face of negative safety studies for tobacco products (see my other Substack);
34) Rickettsia prowazekii vaccine subterfuge and smuggling were used by a couple of scientists forced into the Nazi regime to undermine their war efforts125 126:
Weigl both provided weaker vaccines to the Stasi and smuggled effective ones to the resistance in the Polish ghettos
Fleck organized an elaborate ruse within his lab to provide fake vaccines to the Nazis and if they were questioned, he would supply them with a real sample to quell their suspicions about it not working
in the 1930s and 40s Japanese military scientists conducted biologic warfare research with field and human testing in northeastern China that included the same organism against which these vaccines were made;127
evidence that the intersection of warfare with infectious disease and immunization has a long history
35) ignoring the value of the tenth man strategy- “If there are 10 people in a room and nine agree, the role of the tenth is to disagree and point out flaws in whatever decision the group has reached.”128 Originated with Israeli intelligence after the Yom Kippur war in 1973; or how about the longstanding concept in medicine of a second opinion;
36) the credit reporting/tracking agency Experian verifies user identity for state vaccine verification portals, example Vax Verify in Illinois launched on Aug. 11, 2021: “you are providing “written instructions” to the State of Illinois under the Fair Credit Reporting Act, authorizing the State of Illinois to obtain information from your personal credit profile or other information from Experian.”129 130 Laying the groundwork for social credit score like is in China.
37) the list goes on, but you get the point, right?
Conclusions:
“Ending the pandemic” is not as scientifically realistic in the way that it was portrayed. The actual science points to the fact that while the pandemic may subside, progression to an endemic or periodically epidemic pattern of infection is overwhelmingly likely, and always was. Studies will continue to investigate the durability of immunity in both the vaccinated and those that have recovered from the infection, but it is irrefutable that herd immunity similar to smallpox, measles, and polio for SARS-CoV-2 is, and always has been, an illusion.
The vast majority of prior SARS and MERS vaccine candidates had significant limitations, still unresolved at the time we rolled out the COVID-19 vaccines on the grandest scale imaginable. Taken together, while some progress may have been made in identifying the ability to generate neutralizing antibodies and which might predispose to weaker immune responses or vaccine associated enhanced disease, the findings were conflicting among vaccine platforms, antigen used, animal model type, protection upon viral challenge, and study length. Correlates of protection were still not well established. Animal models cannot recreate the pathophysiology of human infection. Exceedingly few studies assessed protection in aged animals, a crucial information gap since SARS, MERS, and now SARS-CoV-2 all disproportionately affect the elderly and those with certain co-morbidities most severely. Overall the body of knowledge was inconclusive at best regarding the utility of such vaccines during a pandemic and their potential for possible adverse outcomes such as VADE. In fact many articles concluded that application to humans only should be done with caution.
Rushing these vaccines through development and initial testing, then dispensing them worldwide to billions is the opposite of cautious. Especially for an infectious disease with a relatively low fatality rate whose severity is so predominantly focused on a specifically identifiable demographic.
From focused research into basic principles of virology, immunology, and epidemiology combined with research into previous vaccine development and details from their own clinical trial data, the discrepancy between what they were saying publicly and what must have been known from decades of their earned “expertise” is glaring. Shocking even. Pulling the suspicious thread of this one topic (herd immunity) led to uncovering more suspicious threads. Even if the aims were mostly noble, the lies in order to carry them out are deplorable. Tactics have been the very definition of propaganda, censorship, and coercion.
During this timeframe I witnessed the censorship of Google, Twitter, and “mainstream” media evolve. In July my searches began by Google generating results very relevant to my queries. Then late-July/early-August hit. One week it had been search engine as usual, and the very next, the prioritization of results had completely changed. Articles and videos I had previously found at the top of the list were buried beyond page 1. Some no longer to be found. And curiously, the top-hit results now were less academic in kind and now almost exclusively with the words “Fact check” in the top line. I had not changed my search terms. They changed their results to lead people away from established science or discourse on these topics. Additionally, the censorship and attack of those who rightly questioned the narrative became obvious. And they were censoring the exact details that I was finding in virology textbooks and prior SARS-CoV research. Because I happened to be looking right when and where this was occurring, I saw it unfold. This was perhaps the reddest of red flags. If they were so correct and with only pure motives, what did they have to hide?
For supposed “experts” to maintain their original statements in the public eye, and so unequivocally and arrogantly smack down questions, comments, or facts relating to anything possibly in opposition of their narrative, they either had to be criminally incompetent or lying.
If this is “following the science,” what else were they so wrong about? Or worse, lying about?
Welcome to Wonderland.
Johns Hopkins University Center for Systems Science and Engineering (JHU CSSE). Accessed online Oct. 20, 2021. https://github.com/CSSEGISandData/COVID-19.
Public Health England. COVID-19 Vaccine Surveillance Report Week 39. Sept. 30, 2021. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1022238/Vaccine_surveillance_report_-_week_39.pdf
Public Health England. COVID-19 Vaccine Surveillance Report Week 49. Dec. 9, 2021. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1039677/Vaccine_surveillance_report_-_week_49.pdf
Public Health Scotland COVID-19 Statistical Report As at 11 October 2021. Published 13 October, 2021. Accessed online 10/2021. https://publichealthscotland.scot/media/9648/21-10-13-covid19-publication_report.pdf
The Expose. Vaccinated people accounted for 82% of Covid-19 deaths, 69% of hospitalisations & 54% of cases in September according to official figures. Accessed online 10/2021. https://theexpose.uk/2021/10/14/82-percent-covid-deaths-vaccinated-ade/
Shitrit Pnina, Zuckerman Neta S, Mor Orna, Gottesman Bat-Sheva, Chowers Michal. Nosocomial outbreak caused by the SARS-CoV-2 Delta variant in a highly vaccinated population, Israel, July 2021. Euro Surveill. 2021;26(39):pii=2100822. https://doi.org/10.2807/1560-7917.ES.2021.26.39.2100822. https://www.eurosurveillance.org/content/10.2807/1560-7917.ES.2021.26.39.2100822
CDC. Weekly Updates by Select Demographic and Geographic Characteristics. Accessed online 10/2021. https://www.cdc.gov/nchs/nvss/vsrr/covid_weekly/index.htm#Comorbidities
CDC. Weekly Updates by Select Demographic and Geographic Characteristics. From Wayback Machine Feb. 4, 2021. http://web.archive.org/web/20210204003109/https://www.cdc.gov/nchs/nvss/vsrr/covid_weekly/index.htm#Comorbidities
CDC. COVID-19 Mortality Overview. Accessed online 10/2021. https://www.cdc.gov/nchs/covid19/mortality-overview.htm
Woolford SJ, Sidell M, Li X, et al. Changes in Body Mass Index Among Children and Adolescents During the COVID-19 Pandemic. JAMA. 2021;326(14):1434–1436. doi:10.1001/jama.2021.15036. https://jamanetwork.com/journals/jama/fullarticle/2783690
Mahnken K. ‘Staggering’: New Research Shows that Child Obesity Has Soared During Pandemic. The 74. Accessed online 10/2021. https://www.the74million.org/staggering-new-research-shows-that-child-obesity-has-soared-during-pandemic/
FDA. Vaccines and Related Biological Products Advisory Committee October 22, 2020 Meeting Presentation. Accessed online 9/2021. https://www.fda.gov/media/143557/download https://www.fda.gov/media/143557/download
Joint Artificial Intelligence Center of Department of Defense. Humetrix Project Salus: Effectiveness of mRNA COVID-19 Vaccines Against the Delta Variant Among 5.6M Medicare Beneficiaries 65 Years and Older- Weekly update of September 28, 2021. Accessed online 10/2021. https://www.naturalnews.com/files/Salus_Humetrix_VE_study_2021_09_28.pdf
McMorrow M. Improving communications around vaccine breakthrough and vaccine effectiveness. July 29, 2021. https://context-cdn.washingtonpost.com/notes/prod/default/documents/8a726408-07bd-46bd-a945-3af0ae2f3c37/note/57c98604-3b54-44f0-8b44-b148d8f75165.
Pavlich, Katie. Former CDC Director Gives Alarming Statistics on Fully Vaccinated COVID Deaths. Townhall. October 19, 2021. https://townhall.com/tipsheet/katiepavlich/2021/10/19/former-cdc-director-gives-alarming-statistic-on-fully-vaccinated-covid-deaths-n2597628
Page, G. 76% of September Covid-19 deaths are vax breakthroughs. Vermont Daily Chronicle. September 30, 2021. Accessed online 10/2021. https://vermontdailychronicle.com/2021/09/30/76-of-september-covid-19-deaths-are-vaxxed-breakthroughs/comment-page-1/
COVID-19 Vaccine Breakthrough Infections Reported to CDC — United States, January 1–April 30, 2021. MMWR Morb Mortal Wkly Rep 2021;70:792–793. DOI: http://dx.doi.org/10.15585/mmwr.mm7021e3. https://www.cdc.gov/mmwr/volumes/70/wr/mm7021e3.htm
Eric Kowarz, Lea Krutzke, Jenny Reis et al. “Vaccine-Induced Covid-19 Mimicry” Syndrome:Splice reactions within the SARS-CoV-2 Spike open reading frame result in Spike protein variants that may cause thromboembolic events in patients immunized with vector-based vaccines, 26 May 2021, PREPRINT (Version 1) available at Research Square [https://doi.org/10.21203/rs.3.rs-558954/v1] https://www.researchsquare.com/article/rs-558954/v1
Huiru Wang, Qiuchi Chen, Yue Hu et al. Pathogenic antibodies induced by spike proteins of COVID-19 and SARS-CoV viruses, 16 June 2021, PREPRINT (Version 2) available at Research Square [https://doi.org/10.21203/rs.3.rs-612103/v2]. https://www.researchsquare.com/article/rs-612103/v2
Elisa Avolio, Michele Carrabba, Rachel Milligan, Maia Kavanagh Williamson, Antonio P Beltrami, Kapil Gupta, Karen T Elvers, Monica Gamez, Rebecca Foster, Kathleen Gillespie, Fergus Hamilton, David Arnold, Imre Berger, Massimo Caputo, Andrew D Davidson, Darryl Hill, Paolo Madeddu. The SARS-CoV-2 Spike protein disrupts human cardiac pericytes function through CD147-receptor-mediated signalling: a potential non-infective mechanism of COVID-19 microvascular disease. bioRxiv 2020.12.21.423721; PREPRINT available at: doi: https://doi.org/10.1101/2020.12.21.423721. https://www.biorxiv.org/content/10.1101/2020.12.21.423721v2
Liu L, Yuen K-Y, Chen Z et al. Anti-spike IgG causes severe acute lung injury by skewing macrophage responses during acute SARS-CoV infection. JCI Insight. 2019;4(4):e123158. https://doi.org/10.1172/jci.insight.123158. https://insight.jci.org/articles/view/123158
Su, S., Du, L. & Jiang, S. Learning from the past: development of safe and effective COVID-19 vaccines. Nat Rev Microbiol 19, 211–219 (2021). https://doi.org/10.1038/s41579-020-00462-y
Huiru Wang, Qiuchi Chen, Yue Hu et al. Pathogenic antibodies induced by spike proteins of COVID-19 and SARS-CoV viruses, 16 June 2021, PREPRINT (Version 2) available at Research Square [https://doi.org/10.21203/rs.3.rs-612103/v2]. https://www.researchsquare.com/article/rs-612103/v2
Vojdani A, Vojdani E and Kharrazian D (2021) Reaction of Human Monoclonal Antibodies to SARS-CoV-2 Proteins With Tissue Antigens: Implications for Autoimmune Diseases. Front. Immunol. 11:617089. doi: 10.3389/fimmu.2020.617089. https://www.frontiersin.org/articles/10.3389/fimmu.2020.617089/full
Karami Fath M, Jahangiri A, Ganji M, Sefid F, Payandeh Z, Hashemi ZS, Pourzardosht N, Hessami A, Mard-Soltani M, Zakeri A, Rahbar MR and Khalili S (2021) SARS-CoV-2 Proteome Harbors Peptides Which Are Able to Trigger Autoimmunity Responses: Implications for Infection, Vaccination, and Population Coverage. Front. Immunol. 12:705772. doi: 10.3389/fimmu.2021.705772. https://www.frontiersin.org/articles/10.3389/fimmu.2021.705772/full#B80
Lyons-Weiler J. Pathogenic priming likely contributes to serious and critical illness and mortality in COVID-19 via autoimmunity. J Transl Autoimmun. 2020 Apr 9;3:100051. doi: 10.1016/j.jtauto.2020.100051. PMID: 32292901; PMCID: PMC7142689. https://pubmed.ncbi.nlm.nih.gov/32292901/
Schmeck Jr, HM. Swine flu program is halted in nine states as 3 die after shots. New York Times. October 13, 1976. Accessed online 10/2012. https://www.nytimes.com/1976/10/13/archives/swine-flu-prograrm-is-halted-in-9-states-as-3-die-after-shots.html
Fisher R. The fiasco of the 1976 swine flu. BBC online. Sept. 21, 2020. Accessed online 10/2021. https://www.bbc.com/future/article/20200918-the-fiasco-of-the-us-swine-flu-affair-of-1976
Kim HW, Canchola JG, Brandt CD, Pyles G, Chanock RM, Jensen K, Parrott RH. Respiratory Syncytial Virus Disease in infants despite prior administration of antigenic inactivated vaccine. American Journal of Epidemiology, Volume 89, Issue 4, April 1969, Pages 422–434, https://doi.org/10.1093/oxfordjournals.aje.a120955
Aiello A, Farzaneh F, Candore G, Caruso C, Davinelli S, Gambino CM, Ligotti ME, Zareian N and Accardi G (2019) Immunosenescence and Its Hallmarks: How to Oppose Aging Strategically? A Review of Potential Options for Therapeutic Intervention. Front. Immunol. 10:2247. doi: 10.3389/fimmu.2019.02247. https://www.frontiersin.org/articles/10.3389/fimmu.2019.02247/full
Sherman AC, Mehta A, Dickert NW, Anderson EJ, Rouphael N. The Future of Flu: A Review of the Human Challenge Model and Systems Biology for Advancement of Influenza Vaccinology. Front Cell Infect Microbiol. 2019;9:107. Published 2019 Apr 17. doi:10.3389/fcimb.2019.00107 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6489464/
de Wit E, van Doremalen N, Falzarano D, Munster VJ. SARS and MERS: recent insights into emerging coronaviruses. Nat Rev Microbiol. 2016;14(8):523-534. doi:10.1038/nrmicro.2016.81 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7097822/
Pulendran B, Ahmed R. Immunological mechanisms of vaccination. Nat Immunol. 2011;12(6):509-517. doi:10.1038/ni.2039 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3253344/
McElhaney JE, Xie D, Hager WD, Barry MB, Wang Y, Kleppinger A, Ewen C, Kane KP, Bleackley RC. T cell responses are better correlates of vaccine protection in the elderly. J Immunol. 2006 May 15;176(10):6333-9. doi: 10.4049/jimmunol.176.10.6333. PMID: 16670345. https://pubmed.ncbi.nlm.nih.gov/16670345/
Munoz FM, Cramer JP, Dekker CL, et al. Vaccine-associated enhanced disease: Case definition and guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine. 2021;39(22):3053-3066. doi:10.1016/j.vaccine.2021.01.055 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7901381/
Kuldeep Dhama, Khan Sharun, Ruchi Tiwari, Maryam Dadar, Yashpal Singh Malik, Karam Pal Singh & Wanpen Chaicumpa (2020) COVID-19, an emerging coronavirus infection: advances and prospects in designing and developing vaccines, immunotherapeutics, and therapeutics, Human Vaccines & Immunotherapeutics, 16:6, 1232-1238, https://www.tandfonline.com/doi/full/10.1080/21645515.2020.1735227
Hacisuleyman E; Hale C; Saito Y; Blachere NE; Bergh M; Conlon EG; Schaefer-Babajew DJ; DaSilva J; Muecksch F; Gaebler C; Lifton R; Nussenzweig MC; Hatziioannou T; Bieniasz PD; and Darnell RB. Vaccine Breakthrough Infections with SARS-CoV-2 Variants. N Engl J Med 2021; 384:2212-2218. DOI: 10.1056/NEJMoa2105000 https://www.nejm.org/doi/full/10.1056/NEJMoa2105000
Boodhoo, N., Gurung, A., Sharif, S. et al. Marek’s disease in chickens: a review with focus on immunology. Vet Res 47, 119 (2016). https://doi.org/10.1186/s13567-016-0404-3
Yong, E. Leaky Vaccines Enhance Spread of Deadlier Chicken Viruses. National Geographic. July 27, 2015. https://www.nationalgeographic.com/science/article/leaky-vaccines-enhance-spread-of-deadlier-chicken-viruses
Read AF, Baigent SJ, Powers C, Kgosana LB, Blackwell L, Smith LP, Kennedy DA, Walkden-Brown SW, Nair VK. Imperfect Vaccination Can Enhance the Transmission of Highly Virulent Pathogens. PLoS Biol. 2015 Jul 27;13(7):e1002198. doi: 10.1371/journal.pbio.1002198. PMID: 26214839; PMCID: PMC4516275. https://pubmed.ncbi.nlm.nih.gov/26214839/
Mabey DC, Hu V, Bailey RL, Burton MJ, Holland MJ. Towards a safe and effective chlamydial vaccine: lessons from the eye. Vaccine. 2014;32(14):1572-1578. doi:10.1016/j.vaccine.2013.10.016 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3991328/
Kimble JB, Wymore Brand M, Kaplan BS, Gauger P, Coyle EM, Chilcote K, Khurana S, Vincent AL. Vaccine-associated enhanced respiratory disease following influenza virus infection in ferrets recapitulates the model in pigs. J Virol. 2022 Jan 5:JVI0172521. doi: 10.1128/JVI.01725-21. Epub ahead of print. PMID: 34985999.
Szczepanek, S. M. et al. Vaccination of BALB/c mice with an avirulent Mycoplasma pneumoniae P30 mutant results in disease exacerbation upon challenge with a virulent strain. Infect. Immun. 80, 1007–1014 (2012). https://pubmed.ncbi.nlm.nih.gov/22252865/
Mara, A.B., Gavitt, T.D., Tulman, E.R. et al. Lipid moieties of Mycoplasma pneumoniaelipoproteins are the causative factor of vaccine-enhanced disease. npj Vaccines 5, 31 (2020). https://doi.org/10.1038/s41541-020-0181-x https://www.nature.com/articles/s41541-020-0181-x
Seasonal influenza vaccine and increased risk of pandemic A/H1N1‐related illness: first detection of the association in British Columbia, Canada.
Janjua NZ, Skowronski DM, Hottes TS, Osei W, Adams E, Petric M, Sabaiduc S, Chan T, Mak A, Lem M, Tang P, Patrick DM, De Serres G, Bowering D
Clin Infect Dis. 2010 Nov 1; 51(9):1017-27.
Skowronski D. M., De Serres G., Crowcroft N. S., Janjua N. Z., Boulianne N., Hottes T. S., et al. . (2010). Association between the 2008-09 seasonal influenza vaccine and pandemic H1N1 illness during Spring-Summer 2009: four observational studies from Canada. PLoS Med. 7:e1000258. 10.1371/journal.pmed.1000258
Non-neutralizing antibodies induced by seasonal influenza vaccine prevent, not exacerbate A(H1N1)pdm09 disease.
Kim JH, Reber AJ, Kumar A, Ramos P, Sica G, Music N, Guo Z, Mishina M, Stevens J, York IA, Jacob J, Sambhara S
Sci Rep. 2016 Nov 16; 6():37341.
Smatti MK, Al Thani AA, Yassine HM. Viral-Induced Enhanced Disease Illness. Front Microbiol. 2018;9:2991. Published 2018 Dec 5. doi:10.3389/fmicb.2018.02991
Cross-protection and cross-reactive cytotoxic T cells induced by influenza virus vaccines in mice.
Webster RG, Askonas BA
Eur J Immunol. 1980 May; 10(5):396-401.
Vincent A. L., Lager K. M., Janke B. H., Gramer M. R., Richt J. A. (2008). Failure of protection and enhanced pneumonia with a US H1N2 swine influenza virus in pigs vaccinated with an inactivated classical swine H1N1 vaccine. Vet. Microbiol. 126, 310–323. 10.1016/j.vetmic.2007.07.011
Enhanced pneumonia and disease in pigs vaccinated with an inactivated human-like (δ-cluster) H1N2 vaccine and challenged with pandemic 2009 H1N1 influenza virus.
Gauger PC, Vincent AL, Loving CL, Lager KM, Janke BH, Kehrli ME Jr, Roth JA
Vaccine. 2011 Mar 24; 29(15):2712-9.
Rajão D. S., Loving C. L., Gauger P. C., Kitikoon P., Vincent A. L. (2014). Influenza A virus hemagglutinin protein subunit vaccine elicits vaccine-associated enhanced respiratory disease in pigs. Vaccine 32, 5170–5176. 10.1016/j.vaccine.2014.07.059
Doshi P. Pandemrix vaccine: why was the public not told of early warning signs? BMJ 2018; 362 :k3948 doi:10.1136/bmj.k3948. https://www.bmj.com/content/362/bmj.k3948
Fitzpatrick M. The Cutter Incident: How America's First Polio Vaccine Led to a Growing Vaccine Crisis. J R Soc Med. 2006;99(3):156. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1383764/
Nigrovic LE, Thompson KM. The Lyme vaccine: a cautionary tale. Epidemiol Infect. 2007;135(1):1-8. doi:10.1017/S0950268806007096. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2870557/#:~:text=In%201998%2C%20the%20FDA%20approved,%2Deffects%2C%20and%20declining%20sales.
Original Antigenic Sin: How First Exposure Shapes Lifelong Anti–Influenza Virus Immune Responses
Ali Zhang, Hannah D. Stacey, Caitlin E. Mullarkey, Matthew S. Miller
The Journal of Immunology January 15, 2019, 202 (2) 335-340; DOI: 10.4049/jimmunol.1801149 https://www.jimmunol.org/content/202/2/335
Eric L. Brown and Heather T. Essigmann and David W. Pascual. Original Antigenic Sin: the Downside of Immunological Memory and Implications for COVID-19. ASM Journals. Vol 6, No 2. March 10, 2021. https://journals.asm.org/doi/epub/10.1128/mSphere.00056-21
Marek Petráš & Ivana Králová Lesná (2021): SARS-CoV-2 vaccination in the context of original antigenic sin, Human Vaccines & Immunotherapeutics, DOI: 10.1080/21645515.2021.1949953 https://www.tandfonline.com/doi/pdf/10.1080/21645515.2021.1949953?needAccess=true
Branswell H. Getting a flu shot every year? More may not be better. STAT news. Nov. 11, 2015. Accessed online 10/2021. https://www.statnews.com/2015/11/11/flu-shots-reduce-effectiveness/
Peter J. Kerr, Isabella M. Cattadori, June Liu, Derek G. Sim, Jeff W. Dodds, Jason W. Brooks, Mary J. Kennett, Edward C. Holmes, Andrew F. Read. MYXV has become hyperimmunosuppressive. Proceedings of the National Academy of Sciences Aug 2017, 114 (35) 9397-9402; DOI:10.1073/pnas.1710336114 https://www.pnas.org/content/114/35/9397
Rodrigo de Almeida, Cecília Naomi Nakamura, Marina de Lima Fontes, Elenice Deffune, Sérgio Luis Felisbino, Ramon Kaneno, Wagner José Fávaro, Athanase Billis, Marcel Otavio Cerri, Ana Marisa Fusco-Almeida, Maria José Mendes Giannini & Andrei Moroz (2018) Enhanced immunization techniques to obtain highly specific monoclonal antibodies,mAbs, 10:1, 46-54, DOI: 10.1080/19420862.2017.1331804
Savelkoul HFJ, Ferro VA, Strioga MM, Schijns VEJC. Choice and Design of Adjuvants for Parenteral and Mucosal Vaccines. Vaccines. 2015; 3(1):148-171. https://doi.org/10.3390/vaccines3010148 https://www.mdpi.com/2076-393X/3/1/148/htm
Downing NS, Shah ND, Aminawung JA, et al. Postmarket Safety Events Among Novel Therapeutics Approved by the US Food and Drug Administration Between 2001 and 2010. JAMA. 2017;317(18):1854–1863. doi:10.1001/jama.2017.5150 https://jamanetwork.com/journals/jama/fullarticle/2625319
Walsh, G. Biopharmaceutical benchmarks 2018. Nat Biotechnol 36, 1136–1145 (2018). https://doi.org/10.1038/nbt.4305. https://www.nature.com/articles/nbt.4305#citeas
Chen YC & Meh MK. Biopharmaceuticals. IntechOpen, 2018. https://www.intechopen.com/chapters/62512
Recombinant hepatitis B vaccine and the risk of multiple sclerosis: A prospective study
Miguel A. Hernán, Susan S. Jick, Michael J. Olek, Hershel Jick
Neurology Sep 2004, 63 (5) 838-842; DOI: 10.1212/01.WNL.0000138433.61870.82. https://pubmed.ncbi.nlm.nih.gov/15365133/
Ahmed SS, Volkmuth W, Duca J, Corti L, Pallaoro M, Pezzicoli A, Karle A, Rigat F, Rappuoli R, Narasimhan V, Julkunen I, Vuorela A, Vaarala O, Nohynek H, Pasini FL, Montomoli E, Trombetta C, Adams CM, Rothbard J, Steinman L. Antibodies to influenza nucleoprotein cross-react with human hypocretin receptor 2. Sci Transl Med. 2015 Jul 1;7(294):294ra105. doi: 10.1126/scitranslmed.aab2354. PMID: 26136476. https://pubmed.ncbi.nlm.nih.gov/26136476/
Segal Y, Calabrò M, Kanduc D, Shoenfeld Y. Human papilloma virus and lupus: the virus, the vaccine and the disease. Curr Opin Rheumatol. 2017 Jul;29(4):331-342. doi: 10.1097/BOR.0000000000000398. PMID: 28394823. https://pubmed.ncbi.nlm.nih.gov/28394823/
Heikki Hyöty, Francisco Leon & Mikael Knip (2018) Developing a vaccine for type 1 diabetes by targeting coxsackievirus B, Expert Review of Vaccines, 17:12, 1071-1083, DOI: 10.1080/14760584.2018.1548281 https://www.tandfonline.com/action/showCitFormats?doi=10.1080%2F14760584.2018.1548281
Mary S. Holland, Liability for Vaccine Injury: The United States, the European Union, and the Developing World, 67 Emory L. J. 415 (2018). https://scholarlycommons.law.emory.edu/cgi/viewcontent.cgi?article=1056&context=elj
The United States Department of Justice, Office of Public Affairs. Pfizer to pay $14.5 million for illegal marketing of drug Detrol. Oct. 21, 2011. https://www.justice.gov/opa/pr/pfizer-pay-145-million-illegal-marketing-drug-detrol
Thomas, Katie. Pfizer settles a drug marketing case for $491 million. The New York Times. July 30, 2013. Accessed online 10/2021. https://www.nytimes.com/2013/07/31/business/pfizer-settles-a-drug-marketing-case-for-491-million.html
The United States Department of Justice, Office of Public Affairs. Johnson & Johnson to pay more than $2.2 billion to resolve criminal and civil investigations. Nov. 4, 2013. https://www.justice.gov/opa/pr/johnson-johnson-pay-more-22-billion-resolve-criminal-and-civil-investigations
The United States Department of Justice, Office of Public Affairs. Pharmaceutical giant AstraZeneca to pay $520 million for off-label drug marketing. Apr. 27, 2010. https://www.justice.gov/opa/pr/pharmaceutical-giant-astrazeneca-pay-520-million-label-drug-marketing
Vaccines and Related Biological Products Advisory Committee Meeting December 17, 2020, FDA Briefing Document Moderna COVID-19 Vaccine. https://www.fda.gov/media/144434/download
U.S. Department of Health and Human Services. Emergency Use Authorization of medical products and related authorities Guidance for industry and other stakeholders. January 2017. https://www.fda.gov/media/97321/download
Mulangu S, Dodd LE, Davey RT Jr. et al. PALM Consortium Study Team. A Randomized, Controlled Trial of Ebola Virus Disease Therapeutics. N Engl J Med. 2019 Dec 12;381(24):2293-2303. doi: 10.1056/NEJMoa1910993. Epub 2019 Nov 27. https://www.nejm.org/doi/full/10.1056/nejmoa1910993
Chua K, Conti RM, Becker NV. Assessment of Out-of-Pocket Spending for COVID-19 Hospitalizations in the US in 2020. JAMA Netw Open. 2021;4(10):e2129894. doi:10.1001/jamanetworkopen.2021.29894. https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2785079?utm_source=For_The_Media&utm_medium=referral&utm_campaign=ftm_links&utm_term=101821
Pollard, A.J., Bijker, E.M. A guide to vaccinology: from basic principles to new developments. Nat Rev Immunol 21, 83–100 (2021). https://doi.org/10.1038/s41577-020-00479-7 https://www.nature.com/articles/s41577-020-00479-7#citeas
Global Vaccine Safety Summit- Geneva Switzerland, Dec. 3, 2019. https://rumble.com/vng8jr-episode-236-w.h.o.-is-to-blame.html
Lazarus R. Electronic Support for Public Health - Vaccine Adverse Event Reporting System (ESP:VAERS) - Final Report. (Prepared by Harvard Pilgrim Health Care, Inc. under Grant No. R18 HS017045). Rockville, MD: Agency for Healthcare Research and Quality, 2010. https://digital.ahrq.gov/sites/default/files/docs/publication/r18hs017045-lazarus-final-report-2011.pdf
clinicaltrials.gov
Washington HA. Medical Apartheid: The Dark History of Experimentation on Black Americans from Colonial Times to the Present. Anchor; copyright 2008.